Atomic number of niobium1/13/2024 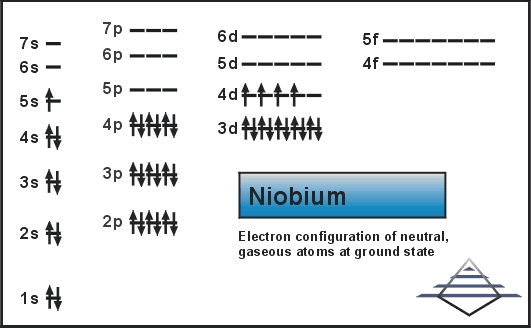
“Niobium alloys and high Temperature Applications” (PDF). Niobium Science & Technology: Proceedings of the International Symposium Niobium 2001 (Orlando, Florida, USA) Cook, “ Development of Columbium Alloy Combinations for Gas Turbine Blade Applications”, (Interim Technical Management Report No. “Development and Production of High Strength Pipeline Steels” (PDF). Niobium Science & Technology: Proceedings of the International Symposium Niobium 2001 (Orlando, Florida, USA). Hillenbrand, Hans-Georg Gräf, Michael Kalwa, Christoph (). “ Naming elements after scientists: an account of a controversy “. Rayner-Canham, Geoff Zheng, Zheng (2008). The most stable radioactive isotope of niobium is niobium-92. This isotope is extremely light weight and is considered as the lightest nuclide and is susceptible to undergo spontaneous fission. Natural niobium has one stable isotope, niobium-93. Inhalation of nitride or pentoxide of niobium in concentration more than 40 mg/m 3 of niobium can lead to severe damage to lungs. When inhaled, it is mainly retained in lungs and bones. It does not react with human tissues which is why it is used in surgical implants. Apart from this, niobium alloys are tested to be physiologically inert and hence harmless. Niobium dust is toxic and is irritant to skin and eyes. Niobium is considered as a potential fire hazard. People rarely are exposed to compounds containing niobium, but some are harmful and should be avoided. Niobium is used in as a catalysts for making acrylic acid.Niobium is used for making wide array of shimmery color jewelry.Niobium is hypoallergenic and is used in making implant devices and prosthetics.Niobium is used to make superconducting magnets for MRI scanners, nuclear magnetic resonance and particle accelerators.Niobium is also used in manufacturing of surface acoustic wave devices, which are essential components of electronic components.Niobium carbide is used in the manufacturing of sharp cutting tools.Nozzle for Apollo Service module was also made using niobium alloy. These alloys show improved strength at low temperatures. Grinders and beams for buildings are also prepared consuming alloys made using niobium. These alloys are used in making rockets and jet engines, oil rigs and gas turbines. Niobium is used to make various super alloys.About 90% of extracted niobium is used in making high grade structural steel. Niobium is used to make special stainless alloys.Niobium is used in making commemorative coins.This allows optical corrective glasses to be made with lighter and thinner lenses. Niobium is used to increase the refractive index of glass.However, niobium reacts with hot concentrated acids and alkalis. It does not react with acids, at room temperature. It readily reacts with carbon, sulfur, nitrogen, and oxygen. Niobium becomes resistant to further corrosion, when a layer of oxide is formed on its surface. Despite being a refractory metal, it has a considerably lower density (i.e. Niobium can produce strong magnetic field. It becomes a superconductor at very low temperatures. It is greyish in color and is changed to blue when exposed to air and oxygen. Niobium is a soft, lustrous, ductile transition metal. It is also found in Australia, Russia and Nigeria. Large deposits of Niobium are found in Brazil and Canada and are the largest producers of niobium mineral concentrates. It is present in abundance at the core or center of the earth. It is ranked as the 33 rd most common element in Earth’s crust. Mineral columbite is the main source of this element. Niobium can be found in nature only in compound state. Later in 1949 at the 15 th Conference of the Union of Chemistry, the name Niobium was chosen as the 41 st element of the periodic table. For almost a century Europe and USA used their own name for this element. Later, due to confusion, it was considered another element, known as tantalum, and in 1846, German Chemist Heinrich Rose proposed that it was indeed a novel element and named it Niobium. Initially, it was named Columbium (with symbol Cb) after Columbia a state of United States. Niobium was discovered in 1801 by Charles Hatchett. It is used in making high-grade structural steel. Niobium was discovered by English chemist Charles Hatchett in 1801, Niobium is a soft, grey, ductile element classified amongst the transition elements of periodic table.
0 Comments
Leave a Reply.AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
