Na element group1/14/2024 The first and second ionization energies are 495.8 kJ/mol and 4562 kJ/mol, respectively. Sodium atoms have 11 electrons, one more than the stable configuration of the noble gas neon. Acute neutron radiation, as from a nuclear criticality accident, converts some of the stable 23Na in human blood to 24Na the neutron radiation dosage of a victim can be calculated by measuring the concentration of 24Na relative to 23Na. Two nuclear isomers have been discovered, the longer-lived one being 24mNa with a half-life of around 20.2 milliseconds. Two radioactive, cosmogenic isotopes are the byproduct of cosmic ray spallation: 22Na has a half-life of 2.6 years and 24Na, a half-life of 15 hours all other isotopes have a half-life of less than one minute. 23Na is created in the carbon-burning process in stars by fusing two carbon atoms together this requires temperatures above 600 megakelvins and a star of at least three solar masses. Twenty isotopes of sodium are known, but only 23Na is stable. Spin-orbit interactions involving the electron in the 3p orbital split the D line into two, at 589.0 and 589.6 nm hyperfine structures involving both orbitals cause many more lines. In a flame test, sodium and its compounds glow yellow because the excited 3s electrons of sodium emit a photon when they fall from 3p to 3s the wavelength of this photon corresponds to the D line at about 589.3 nm. 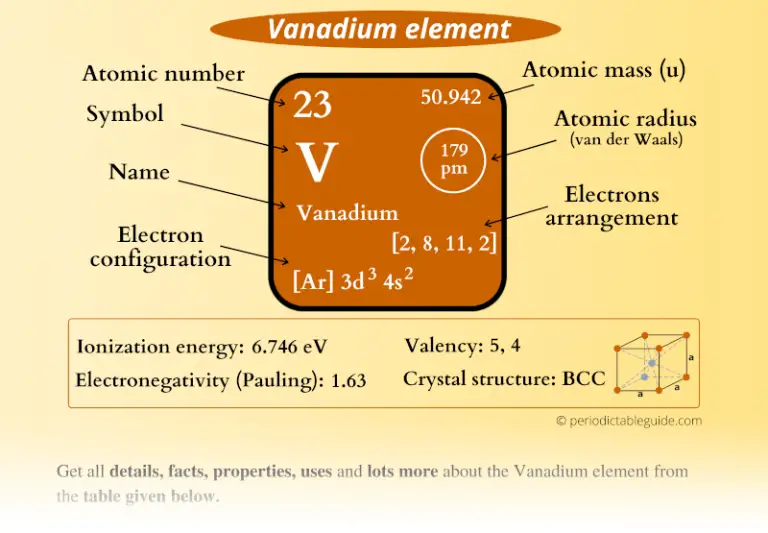
A positive flame test for sodium has a bright yellow color. All of these high-pressure allotropes are insulators and electrides. These properties change dramatically at elevated pressures: at 1.5 Mbar, the color changes from silvery metallic to black at 1.9 Mbar the material becomes transparent with a red color and at 3 Mbar, sodium is a clear and transparent solid. The melting (98 ☌) and boiling (883 ☌) points of sodium are lower than those of lithium but higher than those of the heavier alkali metals potassium, rubidium, and caesium, following periodic trends down the group. Due to having low atomic mass and large atomic radius, sodium is third-least dense of all elemental metals and is one of only three metals that can float on water, the other two being lithium and potassium. It is a good conductor of electricity and heat. Sodium metal can be easily cut with a knife. Bulk sodium is usually stored in oil or an inert gas. Sodium at standard temperature and pressure is a soft silvery metal that combines with oxygen in the air, forming sodium oxides. In nerve cells, the electrical charge across the cell membrane enables transmission of the nerve impulse-an action potential-when the charge is dissipated sodium plays a key role in that activity.Ĭharacteristics Physical Emission spectrum for sodium, showing the D line Isotonic loss of water and sodium from the ECF compartment decreases the size of that compartment in a condition called ECF hypovolemia.īy means of the sodium-potassium pump, living human cells pump three sodium ions out of the cell in exchange for two potassium ions pumped in comparing ion concentrations across the cell membrane, inside to outside, potassium measures about 40:1, and sodium, about 1:10. Loss of water from the ECF compartment increases the sodium concentration, a condition called hypernatremia. Sodium ions are the major cation in the extracellular fluid (ECF) and as such are the major contributor to the ECF osmotic pressure and ECF compartment volume. Sodium is an essential element for all animals and some plants. Among many other useful sodium compounds, sodium hydroxide ( lye) is used in soap manufacture, and sodium chloride ( edible salt) is a de-icing agent and a nutrient for animals including humans.  
Sodium was first isolated by Humphry Davy in 1807 by the electrolysis of sodium hydroxide. Many salts of sodium are highly water-soluble: sodium ions have been leached by the action of water from the Earth's minerals over eons, and thus sodium and chlorine are the most common dissolved elements by weight in the oceans. Sodium is the sixth most abundant element in the Earth's crust and exists in numerous minerals such as feldspars, sodalite, and halite (NaCl). The free metal does not occur in nature and must be prepared from compounds. Sodium is an alkali metal, being in group 1 of the periodic table. It is a soft, silvery-white, highly reactive metal. Sodium is a chemical element it has symbol Na (from Latin natrium) and atomic number 11.
0 Comments
Leave a Reply.AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
